The Practical Application of Genetic Biomarkers in the Individualised Treatment of Breast Cancer
The latest diagnostic and prognostic technology allows clinicians to define appropriate treatment regimens based entirely on the needs and DNA of the individual patient. Choice of breast cancer treatment increasingly depends on your personal mix of gene markers and there is now a new diagnostic test (the EndoPredict) that can provide important information about the way that a tumour is likely to behave at a genetic level.
Breast Cancer Diagnosis and Prognosis - Contents
- Differences between hereditary testing and prognostic tests
- Breast Cancer Prognostic Testing
- Classifying types of breast cancer
- Breast Cancer priority treatment process
- Unnecessary chemotherapy
- Latest breast cancer diagnostic and prognostic tests
- What is the EndoPredict test?
- Identifying types of breast cancer (sub-populations)
Differences between Hereditary Testing and Prognostic Tests
It is important to broadly separate the different types of genetic tests that are available for breast cancer into those that look at hereditary testing and those that are concerned with prognosis. Hereditary cancer testing is most often associated with the breast (and ovarian) cancer genes BRAC1/2. Hereditary cancer gene analysis is normally a blood or saliva test to detect genes such as the BRCA1 or BRCA2 mutation. Knowing the results of these tests may help you and your doctor to be proactive (see Dr James Mackay).
The benefits of hereditary breast cancer screening include:
- Increased surveillance
- Improved outcomes and reduced medical costs through earlier diagnosis
- Counselling patients and family members
- Avoidance of unnecessary interventions for family members who do not test positive
- Inform medical management options
Breast Cancer Prognostic Testing
Although hereditary testing may provide useful information, once a breast cancer tumour is detected further types of genetic testing can inform how it should be treated based on likely prognosis. In these circumstances a biopsy of the tumour is taken and sent to the pathology laboratory (path lab) for histological assessment.
Previously breast cancers were classified according to the cells that they arose from (ductal or lobular) along with other characteristics depending on how the cancer appeared down the microscope (tubular, mucinous etc.). These descriptions said nothing about how the cancer might behave. With increasing understanding of the molecular basis of breast cancer a new classification has arisen that depends more on biological factors, rather than a simple description. This classification not only involves the oestrogen and progesterone receptors (ER and PR) but also the sensitivity of a cancer to the Herceptin drug (Her2) and the Ki67. This is a proliferation index that indicates how fast a cancer is growing and is more sensitive than the “grade” that is often still given.
Classifying Types of Breast Cancer
|
Type of Breast Tumour |
Approximate proportion of all primary breast tumours found |
|
Her2+ - Human epidermal growth factor receptor positive |
10-20% |
|
Triple negative - Human epidermal growth factor receptor negative and do not exhibit oestrogen or progesterone receptors |
15-20% |
|
Luminal type A: ER+ and/or PR+, HER2-, low Ki67 |
40% |
|
Luminal type B: ER+ and/or PR+, HER2+ (or HER2- with high Ki67) |
20% |
Breast Cancer Priority Treatment Process
For early and locally advanced breast cancer the first clinical objective is, usually, to surgically remove the tumour. The next objective is more complex as it requires a decision about the most appropriate form of treatment to reduce the risk of developing a recurrence of the disease in the form of distant metastases. Follow up treatment for the largest proportion of patients who are in the Her2-/ER+ group is challenging as clinicians and patients need to consider the risk/benefit profile when comparing an ‘endocrine only’ or an ‘endocrine + chemotherapy’ regime to reduce the risk of developing distant metastases.
Up until recently most low-risk patients were treated with endocrine therapy only and the high-risk patients with endocrine therapy and chemotherapy. Difficulties arise because many cancers will receive an intermediate prognosis causing significant uncertainty for both the patient and clinician. So, in order to err on the side of caution the majority of these patients received chemotherapy treatment.
In other words these patients can now be treated appropriately without the need for chemotherapy and associated side-effects..
Unnecessary Chemotherapy 
As a result, around half of these ‘intermediate’ patients currently receive unnecessary chemotherapy. Although chemotherapy can be highly effective for some forms of cancer, it is also stressful, has a significant side-effect profile, is toxic by nature and has an impact on mortality. (See Cancer Research UK side-effects of chemotherapy.)
It is therefore highly desirable to be able to identify those patients who would not respond to chemotherapy in any event and to be able to focus care on appropriate treatments including anti-hormonal therapy, instead.
Latest Breast Cancer Diagnostics and Prognostic Test
The latest genetic diagnostic tests combined with an analytical system called EndoPredict means that patients who would not respond to chemotherapy can be spared this treatment and the associated side-effects. These tests are a fundamental move towards providing the necessary individual assessment that will allow a completely bespoke treatment plan for each breast cancer patient, or ‘personalised medicine’. The availability of the latest diagnostic and prognostic tests (in some specialist breast clinics, such as The London Breast Clinic) means that it is now possible to start providing truly individualised breast cancer care.
“It is my view that EndoPredict is the next leap forward in personalising breast cancer care. Its ability to predict the potential for early and late metastases with greater accuracy than any other available test allows chemotherapy to be targeted to those patients who will get the greatest benefit. Equally importantly it can reassure those at low risk of recurrence that chemotherapy is not required. It also represents another step in our understanding of the biology of breast cancer. Such insights are fundamental if the goal of truly individualised treatment is to be realised.”
What is the EndoPredict Test?
Recent studies have shown that the detailed analysis of the growth behaviour of a tumour at the genetic level is an accurate marker of the subsequent actual behaviour of the cancer, as well as what form of treatment the tumour will respond to. These test results now form the basis for the latest recognised medical guidelines.
The EndoPredict test combines genetic markers with other more traditional histology pathology test indicators such as tumour nodal status and tumour size, and provides a major step forward in helping breast cancer specialists to avoid the over and under treatment of breast cancer with chemotherapy. Following sampling, test results are normally available for clinical assessment within a few days.
Identifying types of Breast Cancer (sub-populations)
When breast cancer metastases occur they usually happen within the first three years following the original diagnosis. However, breast cancer tumours positive for hormone receptors (HR+) and negative for HER2/neu can often metastasise at a much later stage. One of the objectives of testing is to calculate the likelihood for recurrence or late metastases.
Most patients with hormone receptor-positive and Her2/neu-negative breast cancer, even if the cancer has infiltrated the lymph nodes have an excellent prognosis. The EndoPredict test identifies those patients who are at low risk for cancer re-occurrence. What this means is that many patients can avoid unnecessary chemotherapy, and have the knowledge that they are sufficiently treated with a five-year anti-hormonal therapy requiring no extended therapy.
Following a core biopsy, EndoPredict examines twelve genes in the tumour cells and identifies those patients who will respond to anti-hormonal treatment, and in more than 95 % of cases will not develop a recurrence within the next ten years. The genes identified are those associated with abnormal cell division (these include UBE2C, BIRC5 and DHCR7), whether or not hormone receptors are expressed (including STC2, AZGP1, IL6ST, RBBP8 and MGP) as well as reference and normalisation genes.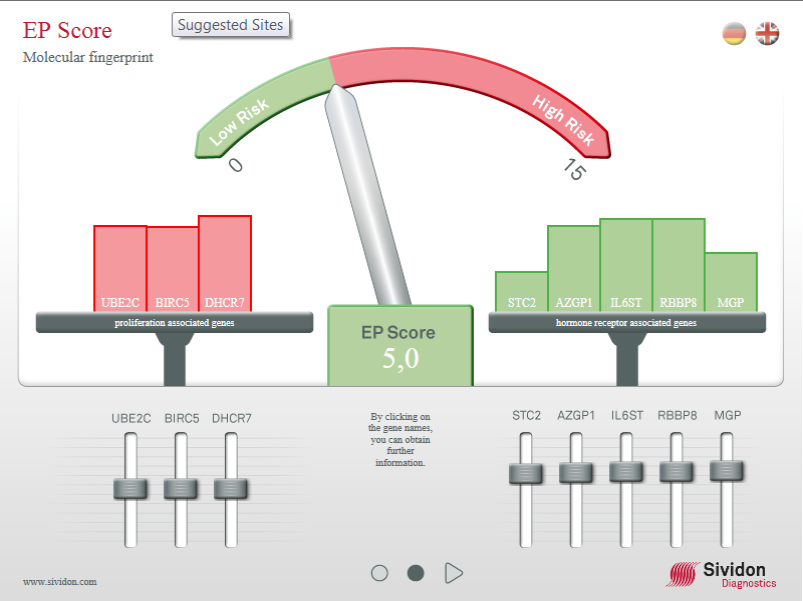
Using this combination means that those 60 % of patients with a low risk profile and eligible for prognosis determination with EndoPredict will be able to avoid chemotherapy. Testing for these genes gives a ‘molecular finger print’ of the patients’ tumour and by combining this information with traditional pathology markers, including nodal status and tumour size, estimates the five and ten-year risk of developing distant metastases.
Links for further information:
- For further information on the EndoPredict testing by Myriad Laboratories
- To request Breast Cancer hereditary testing - Dr James Mackay, Accredited Consultant Clinical Genetic Oncologist
- To request Breast Cancer Prognostic Gene testing - Mr Simon Marsh, Consultant Breast Specialist