This article discusses the symptoms and latest treatments for pancreatic cancer and explains why screening for this life-threatening disease may not be an option.
Contents
- Prognosis of pancreatic cancer
- Symptoms of pancreatic cancer
- Risk factors for pancreatic cancer
- Diagnosis of pancreatic cancer
- Anatomy of the pancreas and surgery
- What can we do when pancreatic cancer is locally advanced?
- FAQs
Prognosis of pancreatic cancer
Pancreatic cancer is the fifth leading cause of cancer-related death in the United Kingdom with more than 8000 new cases diagnosed every year. The prognosis of pancreatic cancer has historically been very poor with approximately 3% of patients alive five years after diagnosis. Surgical resection remains the only potentially curative option and is associated with a 5-year overall survival rate of between 10 and 15%, but can only be performed in less than 20% of cases. This is primarily because the cancer is either too advanced locally, or it has already spread to other organs, particularly to the liver with metastases.
Symptoms of pancreatic cancer
The reason for such a low operability rate is related to the very menacing nature of this cancer, which frequently only becomes obvious when it is too late for surgery. The symptoms are often non-specific and include stomach and back pain, lack of appetite and weight loss; but late onset of diabetes and acute pancreatitis can also be caused by pancreatic cancer. When the tumour is located in the head of the pancreas it can narrow the bile duct and cause jaundice, with yellow discoloration of the skin, dark urine, pale stools and itching.
Risk factors for pancreatic cancer
Several risk factors have been identified in cancer of the pancreas, and smoking remains the strongest link. A recent study demonstrated that approximately one third of pancreatic cancers in the UK in 2010 were caused by smoking. Other risk factors include diabetes, obesity and chronic pancreatitis (frequently caused by excessive alcohol consumption). A weaker association has also been found with Crohn’s Disease, periodontal disease, a diet rich in processed meat and a history of gastric ulcers. People with at least one first-degree relative diagnosed with pancreatic cancer have almost double the risk of someone without a family history of pancreatic cancer.
Diagnosis of pancreatic cancer
Early diagnosis is vital as demonstrated by the correlation between stage of the tumour and survival in the table below. Unfortunately screening is not a feasible option as the incidence of pancreatic cancer is very low compared to other cancers, like breast or bowel cancer. Screening would not be cost effective and it would also require invasive diagnostic modalities, which in themselves carry a risk. To date there is not a national screening programme for pancreatic cancer and prompt investigation of suspicious symptoms remains the best chance for an early diagnosis.
Classification for pancreatic cancer
| Stage | Classification | 5-year survival |
| IA | Tumour ≤ 2 cm limited to the pancreas | 16% |
| IB | Tumour > 2 cm limited to the pancreas | 14% |
| IIA |
Tumour ≤ 2 cm limited to the pancreas Regional lymph node metastasis |
8% |
| IIB |
Tumour limited to the pancreas or spread beyond without involving blood vessels Regional lymph node metastasis |
6% |
| III |
Tumour spread beyond pancreas involving blood vessels With or without regional lymph node metastasis |
3% |
| IV |
Tumour limited to pancreas or spread beyond, with or without involvement of blood vessels With or without regional lymph node metastasis Distant metastasis |
1% |
In general, those patients treated with surgery survived longer, while those not treated with surgery fared worse. However, only about one of every six patients can be treated with surgery.
An abdominal ultrasound and baseline blood tests are usually the first tests requested by GPs but their sensitivity is low. A special blood tumour marker known as CA 19-9, is high in two thirds of patients with pancreatic cancer. A contrast enhanced CT scan is the gold standard pancreatic screening test if a reasonable suspicion exists.
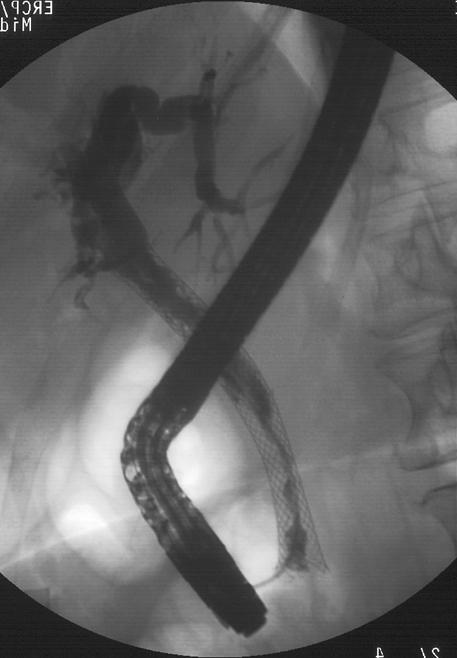
In jaundiced patients, a special MRI scan called Magnetic Resonance Cholangiopancreatography (MRCP) can demonstrate the level of the bile duct obstruction. Most of these patients will require a camera test, called Endoscopic Retrograde Cholangiopancreatography (ERCP), to stent the bile duct (with a plastic or a mesh metal stent) and relieve the jaundice, either as a preliminary step prior to surgery or as definitive treatment in inoperable patients (Figure 1). Cellular material can also be obtained to confirm a cancer diagnosis (known as brushing cytology). There are cases, however, where the diagnosis is uncertain, either because a definite mass cannot be identified on a CT scan or because the features of the lesion are not entirely typical for pancreatic cancer. In these cases a biopsy is recommended and commonly done by Endoscopic Ultrasound (EUS). In this procedure, an endoscopic probe is deployed in the stomach and the tumour is visualised and biopsied. A percutaneous biopsy, usually done by the radiologists under CT or ultrasound scan, should be performed only in patients with clearly inoperable or metastatic disease because of the potential risk of needle track seeding. A Positron Emission Tomography (PET) scan, a nuclear medicine, can be used in selected cases, mainly to clarify metastatic disease such as dubious liver and lung lesions or enlarged lymph nodes.
Anatomy of the pancreas and surgery
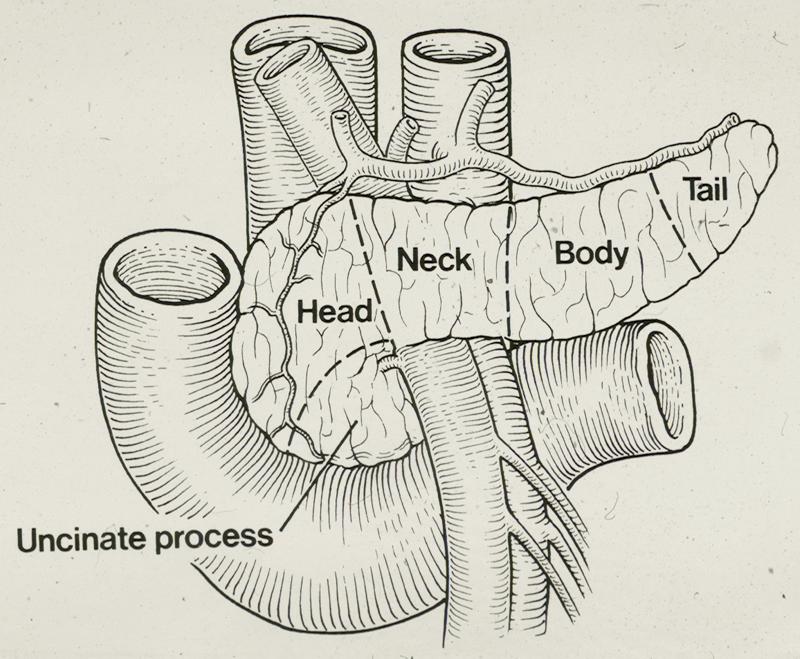
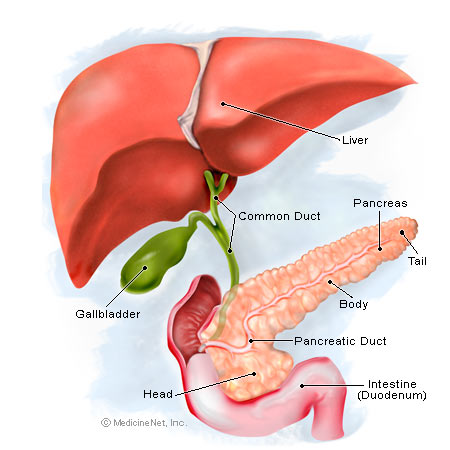
The pancreas is located behind the stomach and is attached to the duodenum, the first part of the intestine in continuity with the stomach, as well as with the bile duct which carries the bile from the liver to the duodenum (Figure 2). The pancreas sits on the two largest blood vessels in the body, the inferior vena cava and the aorta, and the neck of the pancreas is traversed by two important vessels, the superior mesenteric artery and the portal vein (Figure 3).
The pancreas is a gland which produces digestive enzymes (exocrine pancreas) as well as hormones, primarily insulin and glucagon, essential to metabolise sugars (endocrine pancreas). Pancreatic cancer derives from the more common exocrine component. The cells which produce the hormones are scattered throughout the pancreas gland and if they replicate abnormally they can cause a different type of neoplasia called a neuroendocrine tumour. The behaviour of neuroendocrine tumours can be aggressive but very often they are quite benign or even indolent.
Because of its close proximity with vital vascular structures, surgery of the pancreas is particularly delicate and is prohibited in locally advanced tumours which invade some of these vessels.
Pancreato-duodenectomy
Pancreato-duodenectomy is the standard operation for cancer of the pancreatic head. In this procedure, known as the Whipple’s operation, the head of the pancreas is removed together with the duodenum, the bile duct, the gallbladder and the distal part of the stomach. A variant of this procedure adopted by many surgeons is the PPPD, or pylorus preserving pancreato-duodencetomy, where the whole stomach is preserved (Figure 4). Clinical trials have clearly demonstrated no difference in the short- or long-term outcome between the two techniques, though patients undergoing PPPD are more likely to have difficulty in emptying the stomach as the pyloric sphincter is maintained but becomes more prone to developing gastric ulceration and reflux of bile into the stomach.
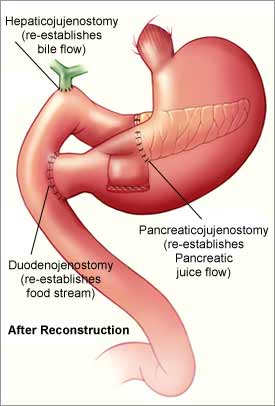
Restoring the continuity of the stomach and bile duct
The continuity of the stomach and bile duct is restored by joining them with a loop of intestine. The pancreatic stump can be joined to the intestine (pancreato-jejunostomy) or to the back of the stomach (pancreato-gastrostomy – Figure 5). Again, there is good clinical evidence that neither of the two techniques is superior to the other and this is purely down to personal preference and experience of the operating surgeon.
Cancer in the tail of the pancreas
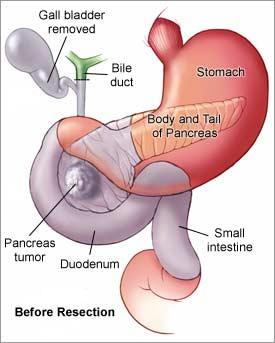
On the other hand, if the cancer is located in the body or tail of the pancreas, the standard operation is a distal pancreatectomy with splenectomy, as the spleen will need to be removed as well for oncological reasons. Laparoscopic or keyhole surgery, sometimes performed for benign or neuroendocrine tumours in the tail of the pancreas, is very rarely considered for cancer, although a few laparoscopic Whipple’s have been reported in specialised centres.
Pancreatic cancer resection
Resection of pancreatic cancer involves major surgery, especially for tumours in the head of the pancreas, as the Whipple’s operation is one of the most invasive and complex surgical procedures. There is a clearly demonstrated correlation between large workload and good results at hospitals and clinics and is a convincing reason to choose high volume centres for this type of surgery.
Patients have to be generally fit to undergo major surgery and will routinely undergo a full pre-operative check-up to assess their cardiorespiratory, renal and nutritional status. Advanced age is not an absolute contraindication to surgery per se and a large number of elderly people are routinely operated on in our unit.
What happens after surgery?
If surgery is uncomplicated patients are expected to be discharged approximately one to two weeks later, although complete recovery can take as long as three months. Improvement in peri-operative care, including anaesthesia and intensive care support, as well as technical progress in surgical and radiological equipment, have led to a drastic decrease in post-operative mortality, with very few patients dying as a result of pancreatic surgery. This improvement is also associated with a better management of the complications, which occur in approximately 30% of cases. The most serious side effects of pancreatic surgery include bleeding, infection and leakage from the pancreas (pancreatic fistula). In some cases where a pancreatic fistula occurs or the gastric function is impaired, a period of intravenous feeding might be necessary to provide nutritional support.
Results from the largest randomised controlled trial, ESPAC-1, have demonstrated a clear benefit in adjuvant chemotherapy after surgery to reduce the chance of recurrence and improve survival. Today the majority of patients are recommended to have gemcitabine post-operatively. The ongoing ESPAC-4 trial is currently recruiting and enrolling patients to compare adjuvant gemcitabine alone versus combination chemotherapy with gemcitabine and capecitabine.
What can we do when pancreatic cancer is locally advanced?
As surgical resection remains the gold standard treatment and the only one to confer potential long-term survival, all efforts should be made to make these patients operable, although several studies have suggested that the long-term survival of patients with residual tumour left behind microscopically (so called R1 resection) can be similar to patients who undergo a curative resection with clear margins (R0 resection), the intention is quite clearly to eradicate the tumour completely. For this reason any intention to debulk a clearly inoperable tumour is not acceptable.
There are other ways to make locally advanced disease operable. Standard downstaging or neo-adjuvant chemotherapy with gemcitabine can shrink the tumour in a relatively small number of cases. However, recently, a new chemotherapy regimen with 5-Fluorouracil, irinotecan and oxaliplatin (folfirinox) has been associated with a response rate as high as 30%. In responsive patients a short course of chemoradiotherapy is usually administered to further decrease the size of the tumour and to reconsider surgical intervention if possible.
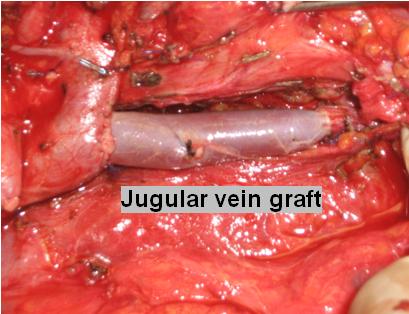
In the absence of metastatic spread, the most common contraindication to operate is the encasement of the arterial vessels. In this case, the tumour is involved in, or even wraps around, the main arterial structures, precluding any surgical treatment, as arterial reconstruction is not a feasible option. On the contrary, if the portal vein is involved, surgery is possible by removing the vein together with the cancer and joining the two ends of the vein together. In cases where the gap is too large to approximate the two ends, the missing part of the portal vein is replaced with a graft, commonly taken from the jugular vein (Figure 6). Several studies have demonstrated that overall survival after portal vein reconstruction is similar to the survival of patients undergoing standard pancreatic surgery and is better than after palliative bypass or other forms of non-surgical palliation. In our Institution, we have extensive experience in treating patients with locally advanced disease and to date we have not recorded any post-operative death after vascular reconstruction of the portal vein in patients undergoing pancreatic surgery.
However, even with the most aggressive and sophisticated techniques, the majority of patients with locally advanced pancreatic cancer remain inoperable. Various treatment modalities have been described in patients with locally advanced and inoperable pancreatic cancer including Cyberknife®, radiofrequency ablation and photodynamic therapy. These techniques utilise different forms of energy in the attempt to locally destroy the tumour but none of them has been proven to be curative and should be regarded as palliative therapies. Cyberknife®, the most promising of these, has been used with some success in selected cases of localised recurrence after surgical resection. In patients with inoperable or recurrent disease, attention should be concentrated on the quality of life, pain relief and the nutritional status of these unfortunate patients, which can be improved with palliative chemotherapy.
FAQs
-
How can pain be controlled and what should be expected immediately after surgery?
Soon after surgery, patients are usually transferred to Intensive or High Dependency Care, where they can be closely monitored for the first 24 hours before they go back to the surgical ward. An epidural, a small anaesthetic pipe inserted on the back of the spine, provides excellent pain relief and is frequently used but some patients prefer to use PCA (patient controlled analgesia), a special device connected to the patient which releases a standard dose of morphine by pressing a button.
-
After surgery, how long do I have to stay in bed and when can I start eating?
Patients are mobilised a few hours after their operation and frequently sit on a chair on the first post-operative day. Although they can usually have some sips of water, proper oral intake is not allowed for the first four or five days.
-
If a complication occurs, is return to the operating theatre always necessary?
Not at all. On the contrary very few patients need surgery to correct a complication. In the majority of the cases complications can be resolved with radiological or simple medical intervention, such as intravenous feeding and antibiotics.
For further information on the author of this article, Consultant Liver Surgeon, Mr Giuseppe Kito Fusai, please click here.
An abnormal proliferation of cells
Full medical glossary



